Research
Background
The immune responses that ensue following vaccination consist of a series of highly orchestrated events involving interactions between B cells, T cells, dendritic cells and follicular dendritic cells in secondary lymphoid organs. The nature of these interactions ultimately dictates the quality and longevity of the immune response generated following vaccination, and each step in the process could potentially be targeted to elicit longer lasting humoral immune responses. The ultimate goal of most – if not all – human vaccines currently in use is to generate antigen-specific long-lived plasma cells (LLPCs). LLPCs provide the host with a persistent source of protective antibodies and are therefore needed to maintain durable immune protection following vaccination. LLPCs and memory B cells (MBCs) are the end products of the germinal center (GC) reaction.
Major Goals:
1) Define the cellular and molecular mechanisms that regulate humoral immune memory generation and maintenance.
2) Examine the heterogeneity of memory B cell responses that are elicited by vaccination? And how do different memory B cell subsets contribute to protection.
3) Determine the rules for eliciting broadly neutralizing B cell responses against rapidly evolving pathogens, such as influenza viruses.
On-going Research
Defining the origin and fate of human B cell responses to influenza virus vaccination.
Previous studies from our group has established that influenza vaccination of humans results in a transient burst of plasmablasts (or antibody secreting cells, ASCs) in peripheral blood (Figure 1). Afterwards, a subsequent wave of activated B cells (ABCs) emerges in blood and seed the memory B cell pool (Figure 1). Therefore, the dynamics and specificity of influenza vaccine induced B cell responses in blood are being thoroughly established. However, there remain major gaps in our current understanding of B cell responses to influenza virus vaccination in humans: (1) does influenza vaccination induce a GC reaction? (2) if yes, are all circulating ABCs GC derived? (3) if no, what is the phenotype of circulating GC derived B cells? 4) which GC-derived antigen-specific B cells are destined to become LLPCs? 5) does influenza vaccination induce a sustained increase in the frequency of bone marrow-resident LLPCs? 6) is there a correlation between the frequency of antigen-specific GC B cells or GC derived circulating ABCs and the increase/maintenance of bone marrow LLPCs? Tackling these gaps will allow us to discern the cellular and molecular determinants that are associated with durable antibody responses to vaccination in humans. In our laboratory, we employ a comprehensive approach that allows us to analyze antigen-specific B cell responses in the blood, draining lymph nodes as well as in the bone marrow after vaccination in humans. We directly and comprehensively interrogate B cell dynamics and the underlying molecular programs through the three major compartments which enable vaccine induced long-lasting humoral immune responses.
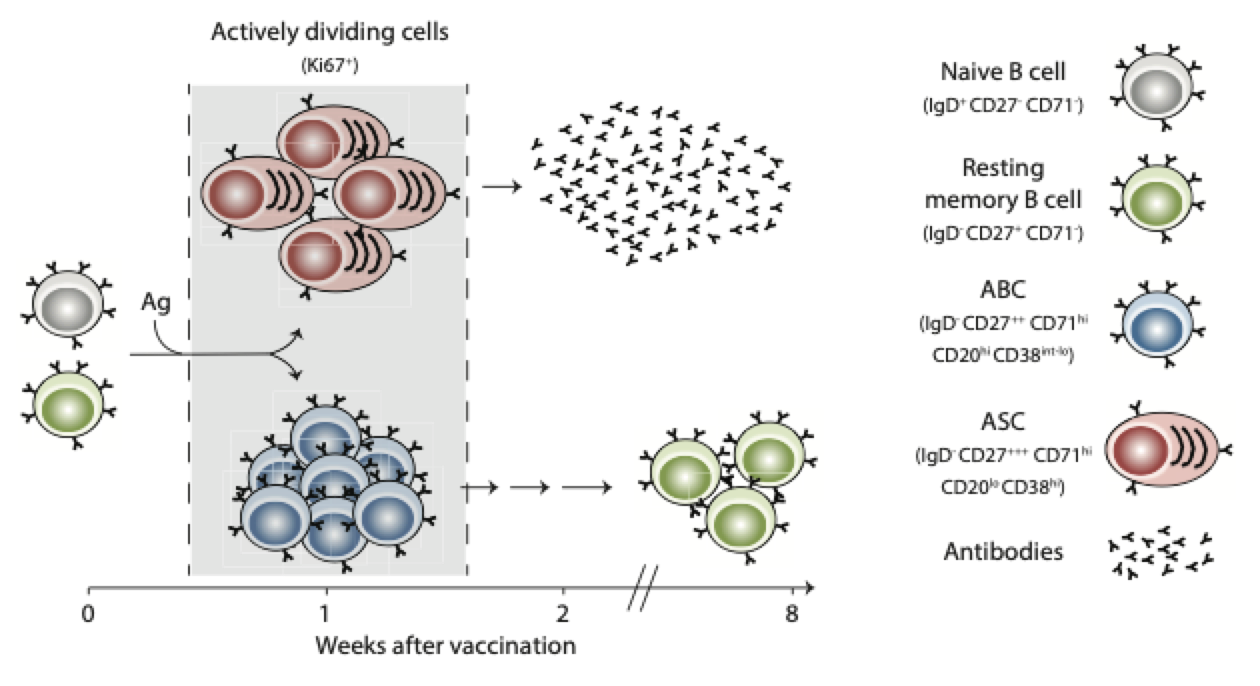
Figure 1. Bifurcation of B cell responses to infection and vaccination in humans (From Ellebedy AH Vaccines 2018)
Rules for eliciting broadly neutralizing B cell responses against influenza viruses.
Some of the most promising approaches to the development of a universal influenza vaccine have come from studies detailing human B cell responses to influenza virus infection and/or vaccination. Such studies have led to the development of a number of broadly reactive human monoclonal antibodies (mAbs) for therapeutic use, the identification of their targeted epitopes, and the creation of immunogens designed to induce the production of such antibodies. With a few notable exceptions, most of these broadly-reactive mAbs bind to the conserved stem region of hemagglutinin (HA). We have defined the HA epitope that is targeted by a broadly neutralizing human recombinant mAb (Figure 2). Our laboratory is actively isolating and characterizing broadly neutralizing anti-influenza virus mAbs that are directed against conserved epitopes within the HA as well as neuraminidase (NA).

Figure 2. Crystal structure of the 701-F02 Fab/ VN/04 HA complex (From Ellebedy AH and colleagues J Virology. 2018 Jul 31)
